Premature Birth
What is a Premature Birth?
How to prevent it and how to respond to it when it occurs
(including newborn care and complications)
Premature birth or preterm birth is said to occur when a baby is born before the completion of 37 weeks of pregnancy. The final weeks of pregnancy are vital for the full development of the baby’s lungs, brain, and liver. Babies born prematurely (especially before 32 weeks gestation) are at higher risk of birth injury including hypoxic-ischemic encephalopathy (HIE) and cerebral palsy.
Low birth weight and preterm birth account for about 17% of infant deaths within the first 12 months after birth. Common complications premature babies may experience include developmental delays, cerebral palsy, breathing problems, and vision or hearing problems—but many of these complications are avoidable. According to the CDC figures, premature birth affects about one out of 10 babies born in the United States.
Medical providers generally use the following terms to describe a premature birth:
- Late preterm: Birth of a baby between 34 and 36 weeks of pregnancy
- Moderately preterm: Birth of a baby between 32 and 34 weeks of pregnancy
- Very preterm: Birth of a baby before the completion of 32 weeks of pregnancy
- Extremely preterm: Birth of a baby before the completion of 25 weeks of pregnancy
Premature Babies Need Special Care
The earlier a preterm birth occurs, the greater is the likelihood of health problems for the baby. While all babies born before the completion of 37 weeks of pregnancy are at a risk of health problems, the likelihood significantly increases when the preterm birth occurs before 34 weeks gestation.
A preterm baby may have to spend time in the NICU (newborn intensive care unit) at a hospital until their vital organs are sufficiently developed to sustain without medical support. NICU care may sometimes be necessary for weeks or months until the baby is:
- (a) ready to breathe without support
- (b) can accept breastfeed or bottle-feed
- (c) has a stable body temperature
- (d) is steadily gaining weight
Even when a premature baby leaves the hospital, they may require medications, special medical equipment, or other medical treatments and support. The medical provider at the hospital may recommend regular checkups from a neonatologist to monitor the baby’s progress.
Health Risks in Premature Babies
Breathing Problems
- Respiratory Distress Syndrome (RDS): In this condition, the baby’s lungs are unable to produce an adequate amount of surfactant (a substance that prevents the collapse of the tiny air sacs in the baby’s lungs.)
- Bronchopulmonary Dysplasia (BPD): Preterm babies can develop this condition, which puts them at an increased risk of contracting lung infections or other types of lung damage.
- Apnea of Prematurity (AOP): This complication in premature babies is said to occur when they experience intermitted pauses in breathing for at least 15 to 20 seconds. AOP may be accompanied by fetal bradycardia or slow heart rate.
Medical Errors and Prevention Strategies
According to researchers preventive as well as therapeutic measures have been well-studied for the underlying causes of breathing problems in premature babies. Timely and correct implementation of neonatal resuscitation measures can reduce the chances of a brain injury from hypoxia or anoxia. Failure to recognize the symptoms and promptly initiate management strategies to address the cause of respiratory distress in babies can result in serious complications. These may include respiratory failure leading to hypoxic-ischemic encephalopathy and cerebral palsy.
The underlying cause of breathing problems in premature babies may not always exist within the lungs alone. After initial neonatal resuscitation and stabilization, the medical providers must evaluate the detailed medical history, conduct a physical examination, and review laboratory and radiographic findings to make a more specific or differential diagnosis and adjust the treatment appropriately.
If the baby shows signs of HIE and meets the criteria, hypothermia treatment should be initiated to slow and/or prevent continuing brain injury.
Intraventricular Hemorrhage (IVH)
IVH is described as a condition when bleeding occurs in the ventricles (fluid-filled spaces) in the baby’s brain. The earlier the preterm birth occurs, the higher is the risk of IVH for the baby. IVH is a major risk factor for cerebral palsy and is seen more commonly in extremely premature babies that have experienced lung collapse, respiratory distress syndrome, or gestational hypertension.

IVH most often occurs within the first 48 hours after birth. Hypoxic-ischemic encephalopathy (HIE) is a serious potential contributing factor to this condition, which can lead to cerebral palsy. Therapeutic hypothermia for preterm babies with a gestational age of 36 weeks or more is the required a standard treatment option in case of moderate to severe HIE that meet certain criteria.
Medical Errors and Prevention Strategies
Intraventricular hemorrhage in premature babies can disrupt the regulatory mechanism of cerebral circulation and result in cerebral hypoperfusion (insufficient blood flow to the brain), leading to secondary brain damage. Ischemia of the white matter (nerve fiber damage) may also occur because of hypoperfusion. Researchers suggest that this condition is reversible if treated early and appropriately.
In order to determine the best treatment, certain imaging tests need to be timely performed. Transcranial Doppler ultrasound can help determine blood flow parameters of cerebral (brain) circulation. With transcranial Doppler sonography, it is possible to perform a detailed blood flow analysis of the cerebral arteries. Using this technique, the medical team can carry out a non-invasive evaluation to determine the grade or severity of brain bleeding in the brain.
Based on the severity of IVH from ultrasound, treatments such as spinal tap to lower the swelling, blood transfusion, and ventilator-assisted breathing should be administered. The higher the grade of brain bleeds, the greater is the risk of HIE and cerebral palsy. In exceptional cases, surgery may have to be performed to correct the cerebral fluid imbalance in the ventricles of the brain. Failure to correctly diagnose or treat IVH can lead to long-term brain damage, developmental delays, and cerebral palsy.
Neonatal Infection and Sepsis
Premature babies are more prone to acquiring infections because their immune system is relatively under-developed. Infection in a preterm baby can result in a severe complication called sepsis, which is the body’s extreme response to infection.
Early onset neonatal sepsis typically occurs during the first week after birth (nine out of 10 cases will present within the first 24 hours of birth.) The most common cause of neonatal sepsis in premature babies is a maternal infection called group B streptococcus that can be effectively treated if diagnosed and managed correctly.
Medical Errors and Prevention Strategies
Research shows that neonatal sepsis continues to be a key cause of serious complications and death in premature and very low birth weight (VLBW) babies. Therefore, medical providers should keep a low threshold for diagnosis and management of sepsis in premature babies. Delayed diagnosis or inadequate treatment is likely to worsen clinical outcomes. The CDC’s national surveillance program from 2005 to 2014 showed that early-onset sepsis (EOS) incidence and infection-related death rate are higher in premature babies.
The ACOG and AAP have updated their guidance for: (a) the use of intrapartum (prior to delivery) antibiotics in expectant mothers with risk of intra-amniotic infection; (b) for intrapartum antibiotic prophylaxis and antenatal screening for group B strep infection; and (c) for antibiotic therapy to preterm babies at risk of early-onset sepsis. However, according to researchers, missed opportunities for the prevention of group B streptococcal infection still continue to adversely affect preterm babies.The medical community has been clear that preventative antibiotics should be liberally used in mothers delivering a premature baby.
Patent Ductus Arteriosus (PDA)
In premature babies, this heart complication may occur when ductus arterious (a blood vessel that allows blood to flow to the baby’s lungs prior to birth) fails to close properly. After birth, the baby’s lungs get filled with air, and the ductus arterious should close on its own within a few days of birth. When this does not occur, excessive blood may flow to the baby’s lungs, causing heart problems.
Medical Errors and Prevention Strategies
When deoxygenated blood in a premature baby passes from the right side of the heart to the left without participating in pulmonary gas exchange (known as excessive pulmonary shunting), PDA diagnosis should be suspected. Chest radiography should be performed to look for signs of pulmonary edema (swelling/inflammation inside the lungs). The amount of blood that the baby’s heart pumps may increase by up to 25% in this condition.
Medical providers should confirm clinical suspicion of Patent Ductus Arteriosus with an echocardiography test. Researchers suggest that preterm babies who are at an increased risk of PDA or whose birth weight is less than 1,000 g should be put on conservative treatment before drug therapy begins. Conservative treatment may include modest fluid restriction and/or use of PEEP (positive end expiratory pressure) to treat pulmonary edema. Instead of using drugs that promote relaxation of the ductus arteriosus, conservative measures may include proactive use of agents such as caffeine that have been associated with lower PDA rates.
But if conservative management is ineffective in controlling pulmonary edema by the second week of birth, drug therapy should be seriously considered. A delay in prescription drug therapy beyond the third week may render the treatment unsuccessful. Early use of NSAIDs, such as indomethacin, within the first one or two days after birth to close a PDA is more effective in reducing the risk of pulmonary hemorrhage than waiting for three to four days after diagnosis.
Necrotizing Enterocolitis (NEC)
This is a fairly common but serious gastrointestinal complication, which can affect a preterm baby’s intestines. Severe inflammation of the intestinal tissue occurs in this condition, causing the death of the tissue. The baby’s intestine may develop a perforation (hole), and harmful bacteria may leak into the bloodstream or the stomach through this perforation. The risk of NEC is 10 times higher in premature babies compared to full term babies.

Recent studies have linked the development of NEC to the ingestion of baby formula during the days and weeks after the birth of premature babies.
Medical Errors and Prevention Strategies
NEC is seen most commonly in premature babies. Research shows that failure to promptly diagnose and treat NEC can result in intestinal stricture (narrowing of the intestine), intestinal perforation, abdominal infection, neonatal sepsis, or even death of the baby. According to researchers, intestinal stricture following NEC often gets misdiagnosed as recurrent constipation, enteritis (intestinal inflammation), or malnutrition, resulting in a higher rate of mortality and morbidity in preterm babies with NEC.
Symptoms of post-NEC intestinal stricture may get overlooked because they appear similar to the clinical presentation of chronic constipation, growth retardation, and malnutrition problems. Therefore, neonataologists, pediatric surgeons, and pediatric gastroenterologists should together follow up the premature baby who is being treated for NEC. A missed NEC diagnosis can result in the baby’s death because of perforation or sepsis.
Retinopathy of Prematurity (ROP)
Premature babies weighing less than 3 pounds at birth are at a risk of developing ROP. This condition occurs with the growth of abnormal blood vessels in the baby’s retina. Some preterm babies will require treatment to prevent blindness from ROP. Eye specialists usually categorize ROP into five stages.

In the first two stages, babies may get better without treatment, but the medical provider must keep a careful watch to see if ROP worsens. In stage three, some babies will require treatment or even surgery to prevent retinal detachment. Babies in stage 4 and 5 will experience partial or complete retinal detachment, and may develop blindness or vision impairment if timely and appropriate surgical intervention is not performed.
Medical Errors and Prevention Strategies
Research shows that retinopathy of prematurity is one of the major causes of avoidable blindness seen exclusively in premature babies, but it can be prevented with timely diagnosis and treatment. While clear guidelines for ROP screening are established, their implementation has been inconsistent by medical providers resulting in higher rates of ROP. Studies have shown that the physician errors and/or their non-availability are the most common causes of ineffective ROP screening.
Researchers suggest that a network of competent screening physicians for ROP in all NICUs should be established. Current guidelines of the AAP and the American Academy of Ophthalmology (AAO) stipulate that all premature babies with a gestational age of less than 30 weeks or a birth weight below 1,500 g should be screened for ROP. Larger babies should also be selectively screened based on medical evaluation.
Gestational age and birth weight are the strongest risk factors for the occurrence of ROP. The ROP screening system should ideally have close to 100% sensitivity so that not a single case of treatment-requiring ROP is missed. Once diagnosed, ROP treatment can include traditional therapy and medications. Laser surgery is a standard treatment for advanced ROP.
Although the FDA has not approved any medications to specifically treat ROP, some drugs approved for other uses may be considered as alternatives to laser surgery or used in combination with it. Avastin (Bevacizumab) drug is FDA approved for colon cancer treatment, but it has also shown promise for ROP treatment, and could be an option for premature babies at the highest risk of blindness. Other FDA approved injectable drugs, such as Lucentis (ranibizumab), Eyelea (aflibercept), and Macugen (pegaptanib) are also being researched and used as ROP treatments.
Neonatal Jaundice
Neonatal jaundice or newborn jaundice is a common condition in premature babies. It occurs when the baby’s liver has not sufficiently developed to eliminate bilirubin (yellow pigment) in the bloodstream. An unusually elevated level of bilirubin may put the baby at the risk of brain damage.

The American Academy of Pediatrics (AAP) recommends that all newborns should be examined for neonatal jaundice at least every 8 to 12 hours while they are in the hospital. Babies must also be examined for jaundice between the 3rd and 7th day after birth because bilirubin levels are likely to peak during this period.
Neonatal jaundice is an extremely treatable condition as long as it is timely diagnosed and aggressive treatment is begun in newborn babies, even premature babies.
Medical Errors and Prevention Strategies
In all newborn babies, elevated bilirubin levels can be potentially neurotoxic leading to brain injury including HIE (the leading cause of cerebral palsy). In particular, preterm babies with this condition will require close monitoring and timely treatment to avoid the development of kernicterus. According to researchers, the use of phototherapy with standard or fiber-optic lights will reduce neonatal jaundice in comparison to no treatment. Neonatal jaundice is generally first visible in the face, and then it progresses to the mid-body and extremities.
It should be emphasized that mere visual estimation of the premature baby’s bilirubin levels without taking blood levels can lead to a misdiagnosis. Medical providers involved in the neonatal care of babies should adopt a low threshold for measuring serum bilirubin by taking a blood. Quicker implementation of phototherapy has been shown to dramatically lower the risk of brain injury and hearing loss.
Sometimes the premature baby will require multiple phototherapy treatment (involving the use of more than one blue light), while the baby is admitted to a newborn nursery or neonatal ICU. In very severe cases, the baby may require a blood transfusion. Failure to accurately diagnose or provide timely and appropriate treatment can quickly worsen the clinical outcomes for premature babies with neonatal jaundice.
Premature Birth and Cerebral Palsy
What does the Research Say?

According to researchers, premature babies account for anywhere between 33% and 50% of all cases of cerebral palsy. Because the premature baby’s brain is under-developed, it increases their risk of HIE that may cause cerebral palsy. A research review of the medical records of 206 children with cerebral palsy showed that 54.4% (more than half) of these children were born prematurely. The highest risk of Cerebral Palsy is associated with babies born before 28 weeks of pregnancy.
Research studies also suggest that preterm birth, particularly before 28 weeks of pregnancy, is a leading risk factor for cerebral palsy. Up to 15% of all babies born between 24 and 27 weeks of pregnancy may develop cerebral palsy. Researchers conclude that antenatal and postnatal care for premature babies should be a critical component of cerebral palsy prevention strategies.
Symptoms and Risk Factors of Preterm Labor
Medical providers and care teams should be alert to the following signs and symptoms that may indicate preterm labor. Early diagnosis can enable obstetricians and nurses to administer treatments to prevent premature birth or take steps to protect the mother and the baby if a premature birth is unavoidable.

Symptoms
- Unusually high vaginal discharge or change in vaginal discharge (bloody, watery or with mucus)
- Complaints of pressure in the lower belly or pelvis
- Persistent dull or low back pain
- Abdominal cramps
- Frequent or regular contractions (with or without pain) in the pre-term period causing abdominal tightness
- Prelabor rupture of membranes (PROM), commonly known as water breaking
- Vaginal bleeding during the second or third trimester, but before the 36th week
Risk Factors
- Previous history of preterm birth
- Family history of preterm birth
- Pregnancy with multiple babies
- Current or past problems with the cervix or uterus
- Diagnosed with certain types of connective tissue disorders
- Preeclampsia (maternal high pressure)
- Gestational diabetes
- Maternal infections, such as group B strep
- Thrombophilia (a condition that increases the risk of abnormal blood clotting)
- ICP or intraheptic cholestatis of pregnancy (a common liver condition that may occur during pregnancy)
- Overweight or underweight before pregnancy, which may also be accompanied by eating disorders, such as bulimia or anorexia
- Getting pregnant again quickly after a previous childbirth
- Getting pregnant after IVF treatment (in vitro fertilization)
- Certain congenital defects are detected in the baby, such as spina bifida (malformation of the spine and spinal cord) or heart defects
- History of excessive stress, smoking, alcohol, or substance use
- Expectant mother’s age is below 17 or above 35
- Expectant mother is of African-American descent (according to the CDC, the rate of preterm birth in African-American women is 14.4%, which is about 50% higher than the rate among white or Hispanic women)
Diagnosis of Preterm Labor

Preterm and premature labor can be diagnosed early when the medical provider (obstetrician, midwife or nurse) is alert to the potential risk factors and symptoms, and orders certain diagnostic tests, such as:
- An ultrasound exam to check the size of the fetus or estimate the gestational age.
- A vaginal swab test to detect the traces of fetal fibronectin. This protein helps ensure that the amniotic sac stays glued to the uterus lining. If the connection between the amniotic sac and the uterus is disrupted, it can result in the release of fetal fibronectin into the secretions around the cervix.
- A pelvic exam or a transvaginal ultrasound exam, which will measure the cervix length to see if it is beginning to thin out and dilate for labor. A transvaginal ultrasound is performed in the vagina, and not on the abdomen. The exam may have to be performed multiple times over a period of several hours.
- If the expectant mother is having uterine contractions prior to 36 weeks, the medical team must monitor them and measure their strength, duration, and frequency. Contractions are monitored using a tocodynamometer, which is a pressure-sensitive device placed over the abdominal area where the contractions are the strongest. Uterine contractions should be monitored along with fetal heart rate monitoring and the recordings should be assessed together and compared.
Prevention and Management of Premature Birth
A number of research-backed medical strategies are available to help prevent or manage the occurrence of preterm labor and premature birth. Medical providers should consider one or more of the following treatment options:
Progesterone Supplementation
For many decades, researchers have recognized progesterone supplementation as a therapy of interest for preventing premature birth. But studies in recent years have provided strong evidence of its efficacy. Progesterone might work because its anti-inflammatory properties could counteract or reverse the inflammatory process involved in the premature initiation of labor.

To prevent premature birth, medical providers should use natural progesterone (instead of synthetic substitutes, which have substantial androgenic activity). The supplementation with natural progesterone may be applied vaginally, by injection, or orally. Vaginal progesterone has minimal side effects. Daily treatment is necessary and doses between 90 and 400 mg may be administered. (No evidence is available to show the superiority of one dose over another.)
Meta analyses of randomized controlled trials show that women with a history of premature birth can reduce their risk of preterm birth in subsequent pregnancies with progesterone supplementation. Considering these findings, ACOG recommends progesterone therapy to prevent recurrent preterm birth. Researchers have also found strong evidence that progesterone supplementation prevents premature birth in women diagnosed with short cervix in mid-pregnancy (from 13 weeks to 27 weeks gestation).
Looking at the usefulness of progesterone in preventing premature birth in women with a short cervix during mid-pregnancy, researchers have pondered whether all expectant mothers should be screened for cervical length during this period. In one of the most significant clinical trials in this area involving a screening of 32,091 asymptomatic expectant mothers, researchers found remarkable results in terms of prevention of birth earlier than 33 weeks.
In relation to this, at this time ACOG does not recommend routine screening of cervical length for all pregnancies, probably because of the large numbers of screenings involved and a perceived need for additional research.
Cervical Pessary

The cervical pessary is a silicone-based soft and flexible medical device. It can be folded and placed around the cervix simply with a vaginal examination and in a completely painless manner. While researchers don’t yet understand the precise mechanism, a cervical pessary may reduce the uterine weight on to the vaginal floor, thereby relieving pressure on the internal os (the narrow opening of the cervix into the uterus).
In this manner, a pessary can help prevent premature cervix dilation and PROM (preterm rupture of the membranes). According to a study published in JAMA, the use of a pessary in women with short cervical length resulted in a lower rate of premature birth earlier than 34 weeks of pregnancy.
Cervical Cerclage
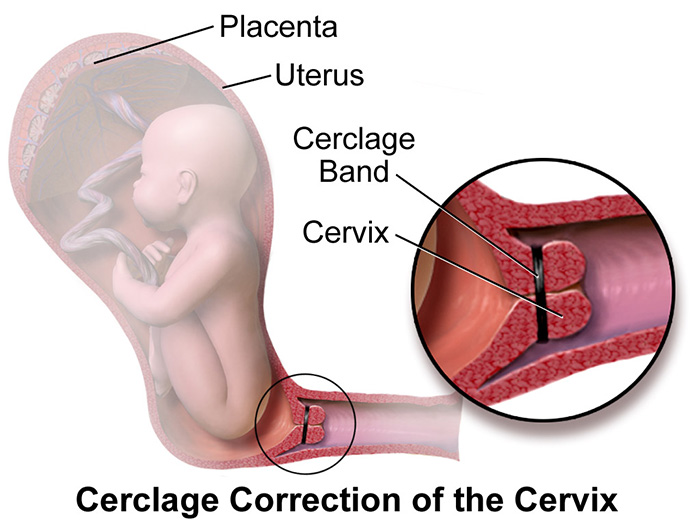
Cervical cerclage refers to the surgical placement of a tape or suture around the cervix with a goal to prevent dilation and resultant premature birth. Medical providers should base their decision to use this treatment in mid-pregnancy after evaluating these 3 factors: (a) History of premature births; (b) Shortened length of the cervix detected with a transvaginal ultrasound test; and (c) Short or dilated cervix detected during a physical examination.
In simplistic terms, cervical cerclage can be described as a physical closure of an incompetent cervix. Meta analyses of randomized clinical trials have shown a reduction of 20% in the rate of preterm births with cervical cerclage compared to no treatment. Research until now has been limited with inconclusive results regarding the superiority of cervical cerclage in comparison to progesterone therapy or cervical pessary.
Treatment of Maternal Infection
Intra-uterine inflammation and infection are known to play a role in the occurrence of spontaneous preterm labor, especially in premature births before 32 weeks of pregnancy or when the pregnancy is complicated by PROM. The vagina is the primary location of maternal infections, and a vigorous inflammatory response to the infection may result in premature birth.
Sexually transmitted infections as well as common vaginal bacteria, such as group B streptococcus and other pathogens may have a role to play in the causal pathway to premature birth. There is some scientific evidence to show that treatment of these infections earlier in the pregnancy can help prevent premature birth. For instance, researchers have shown that administration of an antibiotic medication (such as clindamycin) in expectant mothers with abnormal vaginal microorganisms prior to 22 weeks of pregnancy may lower the risk of premature birth.
With the most common maternal infection, Group B streptococcus, there is research evidence to suggest that premature birth is associated with GBS colonization in expectant mothers, particularly when there are signs of ascending infection (bacteriuria or bacteria in the urine). Although clinical trials have demonstrated the efficacy of intrapartum antibiotic prophylaxis (IAP) in the prevention of neonatal group B strep infections, prevention strategies continue to be inconsistently or inadequately implemented, resulting in continued high rates of group B streptococcal infections in premature babies.
Magnesium Sulfate for Premature Fetal Neuroprotection
In the US, magnesium sulfate is the most widely used drug of choice for the treatment of preterm labor. This compound should only be administered intravenously with an initial infusion of 4g to 6g over 15 to 30 minutes, followed by maintenance doses of 2g to 3g per hour. For expectant mothers who are at a risk of early premature birth (earlier than 32 to 34 weeks of pregnancy), the use of magnesium sulfate for the baby’s neuroprotection may be recommended.

While the drug is being administered, the care team should monitor the mother for clinical signs of magnesium toxicity. The care team should record her pulse rate, blood pressure, rate of respiration, and deep tendon reflexes (such as, patellar – the tendon connecting the kneecap to the shinbone) at least every four hours.
The medical team of doctors and nurses should also closely monitor the health of the baby with electronic fetal monitoring equipment. If there are signs of fetal distress, bradycardia, uterine hyperstimulation or placental abruption the team must act quickly including being prepared to perform an emergency c-section.
Corticosteroids to Support Fetal Lung Maturity
When the medical provider suspects the birth of a premature baby, one of the key steps they can take is to administer corticosteroids (such as betamethasone) to the expectant mother prior to delivery. According to ACOG, during 24 to 34 weeks of pregnancy, a single dose of betamethasone should be administered if the medical team suspects preterm labor to occur within a week.
Antenatal (before birth) steroid treatment is known to accelerate and promote the baby’s lung maturity, reduce their risk of intraventricular hemorrhage, lower the inflammation rates, and shorten the premature baby’s stay in the hospital. The risk of the baby developing respiratory distress syndrome, brain bleeds which could lead to cerebral palsy, white matter damage and other prenatal and postnatal complications is lowered when lungs develop faster with antenatal corticosteroids.
Judicious Use of Tocolytic Drugs to Inhibit Preterm Uterine Contractions
An obstetrical procedure called tocolysis involves the use of medications to delay the birth when an expectant mother presents with preterm contractions. Tocolytic therapy should only be administered for the minimal time possible (up to 48 hours or less in most cases) and promptly withdrawn once the contractions have stopped. After 34 weeks of pregnancy, extending the pregnancy with medications is generally not indicated, considering the risks involved.
Medical providers should individualize the therapy after carefully determining the most effective tocolytic agent for a patient with minimal side effects. Tocolytic drugs used in clinical practice include Betamimetics, Nitric Oxide (NO) donors, Oxytocin receptor antagonists, and Calcium antagonists.
Betamimetics are the most-studied tocolytic medications that inhibit uterine contractions by increasing the intra-cellular concentration of muscle relaxing agents. Pertaining to this, these drugs also have the highest rates of side effects among all tocolytic drugs. Oxytocin antagonists, on the other hand, may have relatively lower maternal and fetal side effects, and could be as effective as betamimetics. Calcium antagonists are the most preferred tocolytic drugs as per the clinical guidelines of the Royal College of Surgeons (UK) because of their efficacy and tolerance.
Is Your Child’s Premature Birth Related Complication the Result of Medical Malpractice?
Parents whose children suffer a birth injury from premature birth related complications deserve answers as to how their child developed this condition and whether the complications resulting from it were preventable. Were there signs during the pregnancy, labor, and delivery process that premature birth or its related complication was occurring, but accurate diagnosis, correct treatment, or an emergency c-section was either delayed or not performed at all leading to birth injuries? Was a neonatal resuscitation team not called to delivery in a timely manner to address breathing difficulties? Were signs of a fetal distress due to preterm birth not recognized and responded to in a timely manner? Did the baby qualify for hypothermia therapy, but the doctors and nurses failed to perform the appropriate analysis or ignored the results of the analysis?
Our dedicated birth injury lawyers want to help you find those answers and obtain that coveted financial reward necessary to provide care and therapy that can help improve the quality of your child’s life. A lawsuit settlement can improve the quality of life for your child.
If your child has been diagnosed with a premature birth injury (including Hypoxic-Ischemic Encephalopathy (HIE) or cerebral palsy), and you suspect this may have been caused in part by medical mistakes, Miller Weisbrod Olesky will thoroughly investigate the facts and hold responsible medical providers and hospitals accountable by pursuing medical malpractice claims against them. The compensation our clients receive helps them pay for their child’s current and future medical treatment, therapy and attendant care to help the child adjust to living with a permanent disability, and to be able to pay for cutting-edge devices that increase a child’s quality of life.
Sometimes families are hesitant to reach out to a medical malpractice lawyer or law firm. It’s also not uncommon for parents to feel overwhelmed by their circumstances and worried that they will not be able to help out in a lawsuit involving their child’s birth injury.
Registered Nurses and Nurse-Attorneys Are a Vital Part of Our Birth Injury Team…and Yours

Most national birth injury law firms will employ one or two nurses to assist the review of cases and medical research. But Miller Weisbrod Olesky offers an unmatched number of nurses and nurse-attorney employees support to both the birth injury attorneys and our clients.
Our team of registered nursing staff and nurse-attorneys bring a deep level of medical and personal insight to every client’s case. Our nursing team includes both an experienced labor and delivery nurse as well as an ICU nurse. Working closely with the rest of the team, they investigate the reasons behind a birth injury and how medical professionals breached their standard of care.
Why Should You Talk with the Knowledgeable Attorneys at Miller Weisbrod Olesky?

The only way to find out if you have a birth injury case is to talk to an attorney who understands birth injuries leading to a delay or failure in developmental milestones including birth injuries that cause cerebral palsy. It’s not uncommon that a premature birth related complication can be a preventable birth injury, but it takes a detailed expert review by a birth injury lawyer of the facts and circumstances of your child’s birth to determine the birth injury was the result of medical malpractice.
At Miller Weisbrod Olesky, a team of committed lawyers, nurses and paraprofessionals uses our detailed medical negligence case review process to assess your potential birth injury case. We start by learning more about you and your child and the status of meeting/missing developmental milestones. Then we gather medical records to determine what happened before, during, and after your delivery. We call in skilled medical experts who review your records and let us know if they think medical errors could have caused your child’s injuries.
If we feel medical negligence caused or contributed to your child’s injuries, we meet with you to discuss how you can receive compensation from the medical professionals who made the errors. Our birth injury attorneys have recovered millions of dollars in settlements for families of babies that have suffered a birth injury.
At no point in our legal intake process will we ask you to pay anything. The medical review of your case and the consultation are free. We only receive payment when you do.
Contact Our National Birth Injury Lawyers
National Birth Injury Law
Our National Birth Injury Attorneys, nurses, and support staff understand that parents of children with birth injuries feel overwhelmed. So, every client has the attention and support of a team of trained, compassionate professionals. But we don’t just offer compassion.
We offer a process to help you discover whether your child’s birth injury, HIE, cerebral palsy or brain injury at birth was caused by medical malpractice.
Call our offices today at (888) 987-0005 for experienced assistance in a free consultation.
Quick Links
Testimonials
- Lyric C. I feel like our voice was heard in a sense of what can possibly go wrong in a delivery and finding us answers. I feel with our settlement, we are now in a comfortable position to provide for our son.
- Lyssa L. They are not just people that say “hey let's get you money and let's go” The law firm was very thorough with us. It was awesome. I don't want to cry, because I think about and it's amazing that they were able to help me and that we were able to help my son and get the story out there.
- Jay C. Throughout the process, one thing was clear to us, the ultimate interest of our child was the utmost concern of Max and his team and as parents navigating a situation like that, that was refreshing to know we had them firmly on our side. I highly recommend them.
Popular Cities
- ★ Dallas Birth Injury
- ★ Houston Birth Injury
- ★ Atlanta Birth Injury
- ★ Chicago Birth Injury
- ★ Philadelphia Birth Injury
Locations
★ Dallas
11551 Forest Central Drive, Ste. 300
Dallas, TX 75243
★ Houston
12929 Gulf Freeway, Ste. 111
Houston, TX 77034